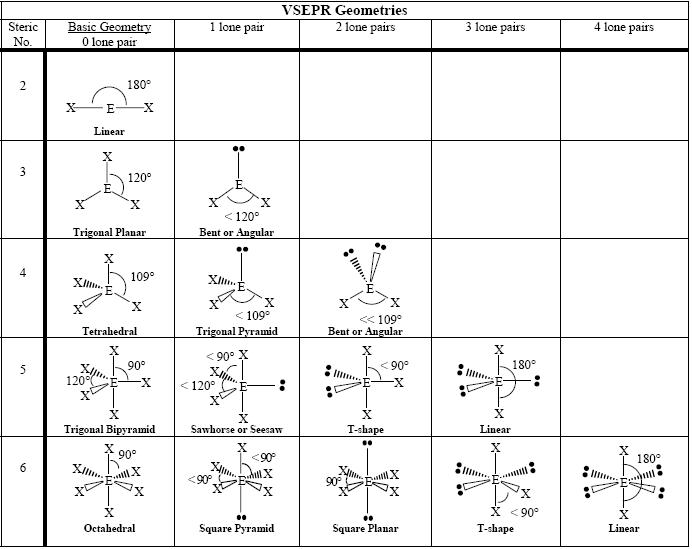
The presence of lone pair causes slight distortion from 109☂8’ to 107☄8’.\) can result from differences in repulsion between various regions of electron density. This shape may either be described as tetrahedral or pyramidal. These four pairs of electrons give rise to a tetrahedral structure where three positions are occupied by H atoms and fourth position by the lone pair. The outer shell then has a share in eight electrons, that is, three pairs bonded and one lone pair. Three electrons of N are bonded with hydrogen and the rest two which do not take part in bonding form the lone pair. Nitrogen is a group 15 element and therefore has 5 electrons in its outmost shell. Q: On the basis of VSEPR theory explain the structure of NH 3 molecule.Īns: In ammonia, N is the central atom. Quantum mechanics and atomic orbitals can give more sophisticated predictions when VSEPR is inadequate. VSEPR also predicts that group-2 halides such as will be linear when they are actually bent.For example, VSEPR predicts that and will have the same bond angles, but structural studies have shown the bonds in the two molecules are different by 12 degrees. First, the idealized bond angles do not always match the measured values.This corresponds to a linear shape with a bond angle of 1 8 0. 
Carbon dioxide is two bonding pairs of electrons and no lone pairs would have the notation A X 2. 5 caused by the increased repulsion from the two sets of lone pairs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
